

Mineralogical studies of ancient ceramics can provide information on the sources of the clay and temperature used at pottery workshops, and can aid in characterising the nature and origin of pottery colour. Such information provides important constraints for the archaeological interpretation of recovered material from ancient localities. In this project we studied pottery from the town of Leptimus, located in modern Tunisia. Although Leptimus has been long established in literary texts as a major exporting centre in North Africa, lack of systematic fieldwork has meant that the town has received only limited recognition from modern scholars. The focus of our study was characterization of the nature of colour changes found in the ancient pottery.
We obtained fragments of amphorae from Leptimus that showed a colour variation in cross section from red on the inside surface to creamy white on the outside surface. The white coating is reported to occur when red firing clays are prepared with sea water, but not with fresh water. For comparison, fragments of modern Tunisian pottery prepared with either sea water (showing a white coating) or fresh water (no white coating) were studied using the same methods. Finally, we also studied some coarse-ware that showed a colour variation from red on the inside surface to black on the outside surface. We used X-ray diffraction, the electron microprobe and Mössbauer spectroscopy to characterise the chemistry and mineralogy of the pottery fragments.
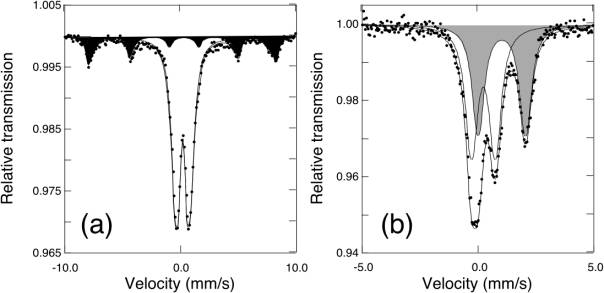
X-ray diffraction showed the crystalline phases in all pottery fragments to be dominated by quartz, and detected no variation between the white or black coatings and the red fired clay. Electron microprobe analyses showed typical calcareous clay compositions with some variation between the coatings and the inside surfaces, but provided no obvious explanation for the colour change. Mössbauer spectroscopy, on the other hand, revealed a simple explanation for the colour changes related to the distribution of iron between the different phases. The red fired clay contained hematite, while the white coatings did not. Additionally, the white coatings showed significant amounts of Fe2+, likely in silicate phases based on the hyperfine parameters (Fig. 3.2-7). The black coatings showed the presence of magnetite. These changes in the distribution of iron are likely too small to be detected by either X-ray diffraction or Mössbauer spectroscopy.
 |
These results are consistent with a previous study conducted on pottery from a different locality, which showed that firing results in migration of iron from oxides such as hematite to calcium silicates and aluminosilicates (mostly pyroxenes and melilites), producing a colour change from red to creamy white. A different study showed that this process is catalysed by the presence of NaCl, which explains why the white coating is most prevalent when the red firing clay is prepared with sea water. The black coating is explained simply as a reduction of Fe3+ in hematite to Fe2+ in magnetite.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page