

Partitioning of Fe and Mg between the lower mantle phases (Mg,Fe)(Si,Al)O3 perovskite and (Mg,Fe)O ferropericlase has been an ongoing topic of research at Bayerisches Geoinstitut, particularly focusing on the determination of separate partition coefficients for Fe2+ and Fe3+ using either Mössbauer spectroscopy or Electron Energy Loss Spectroscopy (EELS). To complement the work being conducted on synthetic samples (see previous article in this Annual Report), a number of natural samples have been studied. These are available as inclusions in diamond, and provide the only means of studying relatively unaltered material from deeper regions of the Earth. The first Mössbauer study on inclusions from the lower mantle was conducted on diamonds from São Luiz, Brazil, and showed that Fe3+ is strongly partitioned into the perovskite phase, while Fe2+ is strongly partitioned into (Mg,Fe)O (see Annual Report 1998). To extend this work to other samples from the lower mantle, we studied a suite of inclusions in diamond from Kankan, Guinea.
Mössbauer spectra of one (Mg,Fe)(Si,Al)O3 and nine (Mg,Fe)O inclusions from Kankan
diamonds were collected at room temperature using the milliprobe technique.
Results for (Mg,Fe)O show relatively low Fe3+/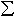 Fe, while the
(Mg,Fe)(Si,Al)O3 inclusion, inferred to have originated from
a perovskite phase, shows significantly higher Fe3+/
Fe, while the
(Mg,Fe)(Si,Al)O3 inclusion, inferred to have originated from
a perovskite phase, shows significantly higher Fe3+/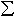 Fe, consistent
with the experimentally-derived relation between Fe3+/
Fe, consistent
with the experimentally-derived relation between Fe3+/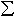 Fe and
Al concentration in silicate perovskite (see Annual Report 1997).
Fe and
Al concentration in silicate perovskite (see Annual Report 1997).
Partitioning of Mg2+, Fe2+ and Fe3+ between (Mg,Fe)O and (Mg,Fe)(Si,Al)O3
was measured directly for one coexisting pair of inclusions, and inferred
for three more coexisting pairs using the experimentally-derived relation
between Fe3+/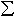 Fe and Al concentration in silicate perovskite
(the silicate perovskite grains in these three diamonds were too small
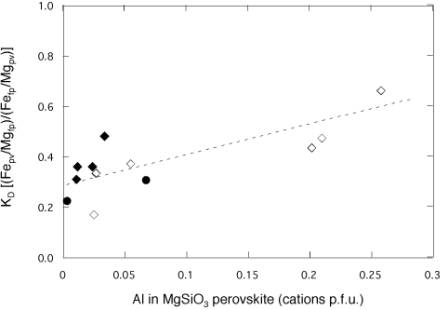
to be studied using Mössbauer spectroscopy). Results for Fetotal/Mg
fall within the range reported for high-pressure experiments on synthetic
samples as well as for other lower mantle inclusions (Fig. 3.3-15), and
show a moderate dependence on Al concentration of the perovskite phase.
More interesting is the dramatic variation of the Fe3+/Mg partition
coefficients between different
Fe and Al concentration in silicate perovskite
(the silicate perovskite grains in these three diamonds were too small
to be studied using Mössbauer spectroscopy). Results for Fetotal/Mg
fall within the range reported for high-pressure experiments on synthetic
samples as well as for other lower mantle inclusions (Fig. 3.3-15), and
show a moderate dependence on Al concentration of the perovskite phase.
More interesting is the dramatic variation of the Fe3+/Mg partition
coefficients between different
 |
samples. One explanation is changes in temperature and/or pressure, since the Fe3+ concentration of (Mg,Fe)(Si,Al)O3 perovskite is known to be at least temperature dependent at constant pressure (see Annual Report 1999). This raises the possibility of defining the first geothermometer or geobarometer for the lower mantle.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page