

The coordination of Ni in silicate glasses and melts has received considerable attention because the abundance, partitioning and structural state of Ni and other transition elements in silicate melts could provide insights into the large-scale geological processes having occurred during the early history of the Earth.
However, the coordination of Ni in silicate glasses is still a matter of considerable controversy. Therefore, in the present study we performed a new investigation of the local environment of Ni in several silicate glasses with different compositions and NiO concentrations using EXAFS spectroscopy.
Pure albite (NaAlSi3O8), diopside (CaMgSi2O6) and sodium disilicate (Na2Si2O5) glasses were prepared by mixing analytical grade oxides and carbonates, melting the mixtures at 1600oC for 1 hour and quenching the melts by dipping the bottom of the Pt cruicible in water. Analytical grade NiO powder was added to the pure glasses and the mixtures were remelted at 1600oC for 1 hour. Eight samples were prepared: albite + 1, 3 and 6 wt% NiO, diopside + 1, 3, and 6 wt% NiO and sodium disilicate with 4 and 12 wt% NiO.
The X-ray absorption experiments were performed at room temperature in transmission geometry on the beamline E4 of DESY at HASYLAB. Data were collected with a double-crystal monochromator using two flat Si(111) crystals. The X-ray intensities were monitored using ionization chambers (IC) filled with mixture of nitrogen and argon gases. Data were collected with energy steps of 5 eV in the pre-edge region (8000 - 8300 eV), 0.3 eV in the edge region (8300 - 8360 eV) and 0.5 eV in the EXAFS region (8360 - 9600 eV). High signal-to-noise ratios were achieved using a 1s counting time per step in the pre-edge region, and 2s in the EXAFS region for small k, which increased further linearly with k, where k is the photoelectron wave vector.
The analysis of the data was performed with the SEDEM suite of programs. The Fourier transform (FT) was performed with Keiser window. The inverse FT of the first peak in the range 0.9 - 2.1 Å, which arises from contributions of oxygen nearest-neighbours, was fitted with the standard EXAFS formula using the amplitude and phase shifts for Ni and O given by McKale et al.
The k3-weighted EXAFS spectra, k3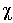 (k), and
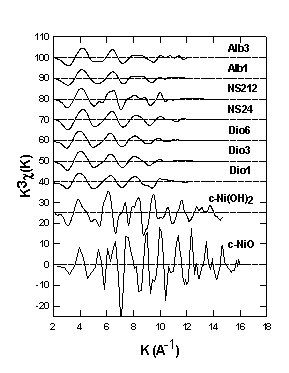
the corresponding phase-uncorrected FT of the investigated samples are
shown in Fig. 3.5-11 and Fig. 3.5-12 respectively. The EXAFS spectra for
NiO and Ni(OH)2 are in excellent agreement with published data.
(k), and
the corresponding phase-uncorrected FT of the investigated samples are
shown in Fig. 3.5-11 and Fig. 3.5-12 respectively. The EXAFS spectra for
NiO and Ni(OH)2 are in excellent agreement with published data.
The preliminary results from the fit of the first shell parameters indicate that: (1) in the diopside and albite glasses the average coordination number of Ni is between 4 and 5. There is no significant change in the first and the second shell around Ni with increasing Ni concentration; (2) in the sodium disilicate glasses the average coordination number of Ni is about 5.5, and is practically independent of composition, some changes in the second shell around Ni with increasing Ni content are observed; which may be due to partial crystallization (3). The average Ni-O distance in all glasses is about 2.00± 0.02 Å, which is intermediate between those expected for four- and six-coordinated Ni. These results will be cross-checked in the future using other EXAFS programs, optical spectroscopy as well as anomalous wide-angle X-ray scattering.
 |
 |

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page