

In the BGI annual report 1996 we presented first results from 1 atmosphere experiments applying the mechanically assisted equilibration technique (MAE technique) to investigate the solubility and partitioning behavior of the highly siderophile elements (HSE) Pt and Rh in silicate melts. These previous results were attained using inductively-coupled-plasma mass spectrometry (Diss.-ICP-MS) to analyse the element concentrations in bulk samples of the experimental glasses. Recently we re-measured these element concentrations by applying laser-ablation techniques (LA-ICP-MS) at ANU, Canberra, Australia. LA-ICP-MS has the advantage of being a spot-analytical technique whereby the influences of micro-nuggets of metal in the glass can be avoided by applying time resolved signal accumulation and data processing techniques.
LA-ICP-MS results for the effect of 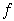 o2 on Pt and Rh solubilities at 1300°C are given in Figure 3.3-5. For oxygen fugacities above -5 log units Rh solubilities are approximately half an order of magnitude higher than those of Pt. The slope of the
o2 on Pt and Rh solubilities at 1300°C are given in Figure 3.3-5. For oxygen fugacities above -5 log units Rh solubilities are approximately half an order of magnitude higher than those of Pt. The slope of the 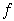 o2 dependence indicates that both Pt and Rh are dissolved in the melt predominately as the oxidised species Pt2+ and Rh2+. At higher
o2 dependence indicates that both Pt and Rh are dissolved in the melt predominately as the oxidised species Pt2+ and Rh2+. At higher 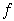 o2 conditions (log
o2 conditions (log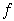 o2 > -1) a change in slope for Rh solubility may indicate that it is present in the melt as Rh3+. At oxygen fugacities below approximately -5 log units the solubilities appear to level out and are reversed with Pt higher than that of Rh. This is due to the appearance of micro-nuggets of metal that form inside the melt samples and bias the analyses. The reversal of the apparent solubility might indicate that Pt tends to form micro-nuggets more easily and/or in higher amounts than Rh.
o2 > -1) a change in slope for Rh solubility may indicate that it is present in the melt as Rh3+. At oxygen fugacities below approximately -5 log units the solubilities appear to level out and are reversed with Pt higher than that of Rh. This is due to the appearance of micro-nuggets of metal that form inside the melt samples and bias the analyses. The reversal of the apparent solubility might indicate that Pt tends to form micro-nuggets more easily and/or in higher amounts than Rh.
Comparison with our previous bulk analysis ICP-MS results shows the clear advantages of applying LA-ICP-MS techniques. Sample ablation was performed in time resolved data acquisition mode. The counts per time interval show direct evidence for micro-nugget contamination as spikes or hills in the count rates. The data processing allows the count rates to be viewed and regions excluded from the final integration. The result of this data procedure is a lower analysed element concentration in the sample, and less scatter in the data themselves.
The final consequence of this analytical progress is reflected in the lowest solubility levels and the highest metal/silicate partition coefficients ever reported for Pt and Rh in a haplobasaltic silicate melt. These results can be extrapolated to lower 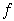 o2 to give an estimate of the partition coefficients for Pt and Rh at conditions believed to be relevant for core formation in the Earth (equivalent to
o2 to give an estimate of the partition coefficients for Pt and Rh at conditions believed to be relevant for core formation in the Earth (equivalent to 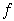 o2
o2 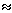 -12 log units at 1 atm and 1300oC). The calculated partition coefficients are extremely large (
-12 log units at 1 atm and 1300oC). The calculated partition coefficients are extremely large (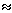 1015 for Pt and
1015 for Pt and  1012 for Rh) and would cause these elements to be almost entirely partitioned into the Earth's core. Non-chondritic element ratios would also result since the partition coefficients of Pt and Rh differ by about 3 orders of magnitude, an order of magnitude more than that observed in Earth's upper mantle. These results, therefore, support the model with addition of a late chondritic veneer to the early Earth after core formation had mainly ceased. The influence of pressure on HSE solubility and partitioning behavior, however, is still an unresolved issue. The latest discovery of micro-nuggets in natural samples gives additional evidence that micro-nuggets may be a natural phenomenon in magmatic systems, forming a kinetically or even thermodynamically stable phase.
1012 for Rh) and would cause these elements to be almost entirely partitioned into the Earth's core. Non-chondritic element ratios would also result since the partition coefficients of Pt and Rh differ by about 3 orders of magnitude, an order of magnitude more than that observed in Earth's upper mantle. These results, therefore, support the model with addition of a late chondritic veneer to the early Earth after core formation had mainly ceased. The influence of pressure on HSE solubility and partitioning behavior, however, is still an unresolved issue. The latest discovery of micro-nuggets in natural samples gives additional evidence that micro-nuggets may be a natural phenomenon in magmatic systems, forming a kinetically or even thermodynamically stable phase.
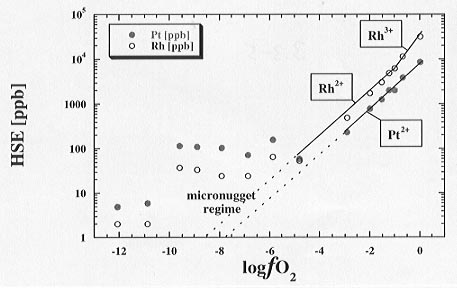 |
Fig. 3.3-5: Solubilities of Pt and Rh in a haplobasaltic silicate melt at 1300°C. Slopes through the solubility results indicate Pt and Rh in divalent oxidation state, with evidence for Rh3+ at the highest fo2 conditions. At low fo2 micro-nugget formation occurs (micro-nugget regime = "apparent solubility"). Solid lines are fits to the data while dashed lines are extrapolations of the curve fits to lower fo2 values. |

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page