

In the past decade numerous crystalline phases have been reported to undergo
a crystalline to amorphous transition as a function of pressure at room
temperature. For quartz and hexagonal GeO2, disordering occurs
at a length scale of less than 100 Å as a result of failure of the
oxygen sublattice to achieve a cubic body-centered symmetry upon densification.
While many studies have focussed on the structural details of pressure-induced
amorphization, nothing about the energetics of this process has yet been
determined. Particularly important in this respect is the comparison of
pressure-amorphized materials with densified glasses. To investigate these
aspects, we have measured enthalpies of solution in hydrofluoric acid of
GeO2 glass and hexagonal crystals compressed statically up to
pressures of 15 GPa in a multi-anvil apparatus. For both kinds of materials,
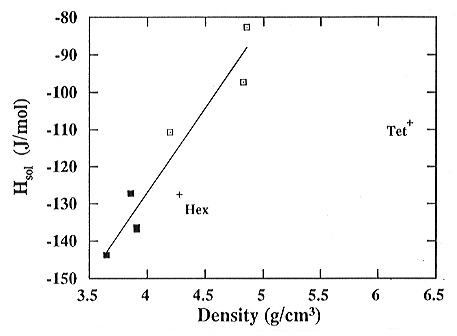
we have found that the same linear relationship obtains between the density
and the enthalpy of solution (Fig. 3.3-5). Increasing the density
 |
|
|
from 3.6 g/cm3 (glass at 1 atm) to 5.0 g/cm3 (crystal amorphized at 15 GPa) leads to an increase of 60 kJ/mol. By comparison, the enthalpy of the transition between the hexagonal and tetragonal forms is only 20 kJ/mol for an increase in the density from 4.3 to 6.3 g/cm3. Consistent with available EXAFS, infrared and Raman data, the measured densities indicate that Ge is essentially four-fold coordinated in the decompressed samples. The energy stored in densified materials through straining of bonds is thus three times as high as that needed for the reconstructive crystalline phase transition.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page