

Despite the low abundance of highly siderophile elements such as nickel and cobalt in the Earth´s mantle, which resulted from the formation of the metallic core, nickel and cobalt place important constraints on the early history of the Earth. Thus the geochemistry of such elements and their partitioning behaviour illustrate the nature and efficiency of processes operative in the early stages of the Earth´s evolution. The direct determination of such partition coefficients at high pressure under well-constrained conditions remains an experimental challenge that is complemented by extrapolations from low pressure partitioning data to higher pressures. Towards this end we are engaged in the determination of the partial molar volumes of NiO and CoO in silicate melts of variable chemistry, which also cast light on the structural role of these elements in melts.
We have therefore measured one-atmosphere densities in air of a number of Ni- and Co-bearing silicate melts using the very precise double-bob Archimedean method. Starting compositions selected were a series of glasses synthesised by the addition of NiO and CoO to sodium disilicate with up to 9.1 mol% NiO and up to 21.4 mol% CoO. Investigations are presently extended to the one-atmosphere diopside-anorthite eutectic system.
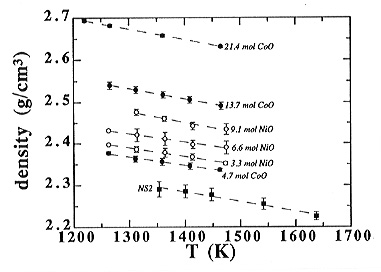
Densities of the Ni- and Co-bearing sodium disilicate melts were determined up to 1473 K and are plotted in Fig. 3.6-5. For the Ni-bearing melts, they range from 2.2 (0% NiO) to 2.5 g/cm3 (9.1% NiO) and for the Co-bearing melts from 2.2 to 2.7 g/cm3, with the highest density corresponding to the most Co-rich melt. Furthermore, the addition of both CoO and NiO results in a nearly parallel shift of the density vs. temperature lines, which suggests that the thermal expansion hardly changes with the addition of these siderophile elements within the temperature range investigated.
In order to investigate the influence of NiO and CoO on the volumetric
properties, an alternative representation of these results is to plot the
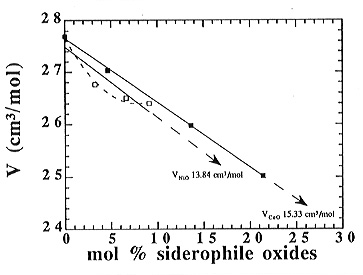
molar volume as a function of the content of siderophile elements, as shown
in Fig. 3.6-6. In this case, the molar volume of Ni-bearing sodium
disilicate melts seems to vary nonlinearly from the sodium disilicate to
the most Ni-rich melt, which may suggest the occurrence of interactions
involving nickel. In contrast, the molar volume of Co-bearing melts decreases
linearly with increasing Co content from the sodium disilicate up to the
most Co-rich melt.
 |
|
|
By fitting the data plotted in Fig. 3.6-6, one can determine the partial
molar volume of NiO and CoO in the composition and temperature range investigated.
The volumetric data of Co-
 |
|
|
bearing melts were fitted linearly, which yields a partial molar volume for CoO of about 15.33 cm3/mol at 1500 K. On the other hand, as previously mentioned, the Ni-bearing melts behave non-ideally with respect to the molar volume. However, the compositional range investigated is not wide enough to justify a polynomial fit, and the preferred linear fit yields a minimum value for the partial molar volume of NiO of about 13.84 cm3/mol at 1500 K. To further explore the suspected nonideality, first attempts to investigate melts with higher Ni- and Co-contents are in progress, but so far the results are ambiguous, possibly due to compositional heterogeneities of these Ni- and Co-rich melts.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page