

A solution to the problem of the "siderophile element anomaly" is one of the keys to understanding the formation of the Earth's core. In the last decades high pressure devices have opened new possibilities to explore this problem by enabling the investigation of element partitioning at elevated pressures. The majority of recent studies have concentrated on whether the influence of pressure is sufficient to solve the "siderophile element anomaly"; however the influence of temperature has been the subject of only a few studies in which other variables may also have affected the results.
Based on the well established result that high pressure can indeed significantly affect the partitioning of siderophile elements (e.g. Ni, Co) between metals and silicate melts or metals and mantle phases, we are studying systematically the influence of temperature. An important goal is to isolate the temperature effect, since it has been shown that other parameters such as pressure, composition and redox conditions can also significantly influence element partitioning.
Experiments between liquid Fe-Ni metal and magnesiowüstite were performed at 9 GPa in the temperature range 1800 to 2400°C using a multianvil apparatus. The starting material was Fe-Ni powder, doped with traces of siderophile elements and tightly packed into MgO capsules. During the experiment the liquid metal reacted with the capsule to form magnesiowüstite solid solutions.
In order to exclude possible effects due to varying oxygen fugacity,
the resulting distribution coefficients DMmet/ox have
been recalculated to a constant oxygen fugacity (log fO2
= -2.7, relative to Fe-FeO). The results are shown in Fig. 3.2-1 and indicate
that distribution
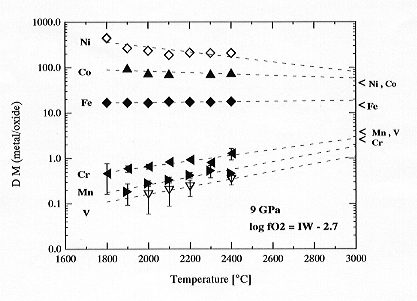 |
Fig. 3.2-1: Metal-magnesiowüstite distribution coefficients as a function of temperature at 9 GPa. The distribution coefficients are recalculated to a constant oxygen fugacity in order to isolate the effect of temperature. The results are extrapolated to higher temperatures (dashed line) and compared to distribution coefficients which are required to support an equilibrium model for the formation of the Earth's core (indicated by arrows). |
coefficients for Ni and Co decrease slightly with temperature, while those for Mn, Cr and V increase slightly. The results for V, however, have very large uncertainties due to low V concentrations that are close to the detection limit of the electron microprobe.
Extrapolation of the results to higher temperatures suggests that the data converge at very high temperatures (> 3000°C) to the DM values that are required to solve the siderophile element problem by equilibrium processes during core formation. The pressure dependence of the DM values are consistent with this suggestion: DNi and DCo decrease significantly with increasing pressure, while the pressure dependence for Mn, Cr, V is negligible. Given that a variation in oxygen fugacity can also change the distribution coefficients, an equilibrium model for the formation of the Earth´s core could be viable. However these elements are only a few of those responsible for the siderophile element problem; consequently more information is required to provide a completely consistent model for the formation of the Earth's core.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page