

Mountain-building events on the earth, a result of the collision of continental and/or oceanic lithospheric plates, cause a disruption in the thermal and gravitational stability of the lithosphere. During collision, as well as the drive back towards an equilibrium state, individual rock particles within the lithosphere may undergo a complex sequence of burial, exhumation, heating and cooling stages. These changes in depth and temperature are partly recorded in the form of mineral assemblages, mineral compositions, and mineral reactions and it is the goal of the metamorphic petrologist to try and reconstruct the temporal, thermal and baric history of a rock unit on the basis of the petrological information contained in the present-day rock sample. The determination of pressures (P) and temperatures (T) - geothermobarometry - requires a knowledge of the equilibrium compositions of mineral phases at a given moment in the rocks' evolution. In reality, due to various kinetic barriers to mineral growth or decay, minerals may be compositionally zoned, records of incompleted reactions may be present, and phases may have survived metastably. Therefore, equilibrium cannot be assumed and the petrologist must evaluate which phases (if any) or, in the case of zoned minerals, which compositions, may have represented a stable assemblage at a given point along the P-T path followed by the rock. Misinterpretation of textures can lead to significantly different P-T results!
A property of garnet, a common mineral in metamorphic rocks of different bulk compositions, is the relatively slow diffusivity of its major elements; changes in conditions during garnet growth are therefore often recorded in the form of chemical zoning. As garnet often also contains inclusions of other mineral phases, it is possible, assuming that the composition of the inclusions and adjacent garnet have not been later modified, to determine a series of P-T points pertaining to the garnet growth history. This method requires that the garnet grew outwards from core to rim - which, as shown below, is not always the case.
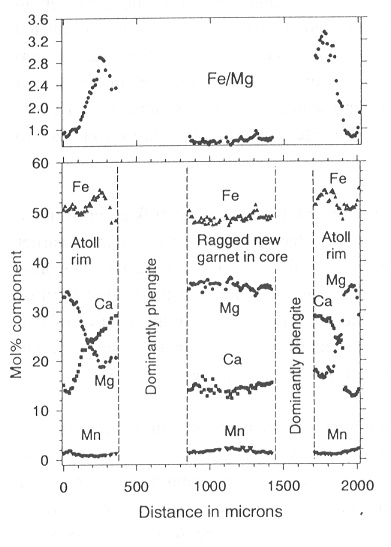
An eclogite studied from the Dabie Shan Mts. of China (in collaboration with D.A. Carswell, Sheffield, U.K.) contains numerous atoll-like garnets. These atoll garnets are cored by large phengite crystals which also contain scattered epidote, omphacite, amphibole, quartz, and rutile inclusions as well as irregular garnet grains. The atoll garnets are strongly zoned (Fig. 3.4-2). From the interior to the rim, Mg climbs steadily at the expense of Ca - core-rim differences of 15 mol% are seen over a distance of 300-500 µm - and Fe/Mg changes from 3.4 in the core to 1.5 at the rim. Interestingly, the ragged garnets inside the phengite have the same composition as garnet rims. If it is assumed that the atoll garnet grew around phengite then the garnet interior composition would have to be combined with the phengite composition to determine temperatures by the garnet-phengite Fe-Mg exchange geothermometer. An alternative explanation is apparent when the rare non-atoll garnets of the same rock are investigated. These also have Ca-rich, Mg-poor cores (Fe/Mg > 7.0) and Mg-rich, Ca-poor (Fe/Mg = 1.5) rims (Fig. 3.4-3) and are packed with inclusions of epidote, omphacite, amphibole, quartz, and rutile; inclusions of phengite have not been observed. The rock matrix contains abundant evidence for the growth of hydrous phases under high pressure conditions in the form of coarse-grained, cross-cutting zoisite, amphibole and phengite. An alternative explanation for the atolls is therefore that the Mg-rich garnet rim composition is the one in equilibrium with the matrix phases and that metastable, Mg-poor, Ca-rich garnet cores have been consumed by zoisite-, phengite- and amphibole-, and ragged Mg-rich garnet-producing reactions during the period of fluid influx. Temperatures calculated based on phengite composition paired with garnet core (high Fe/Mg) and garnet rim (low Fe/Mg) differ by around 250°C: quite a substantial contrast. It is therefore of paramount importance to correctly interpret the growth sequence and textural relationships of natural rocks. Growth is not always from core to rim and breakdown does not necessarily occur only from the outside in!
 |
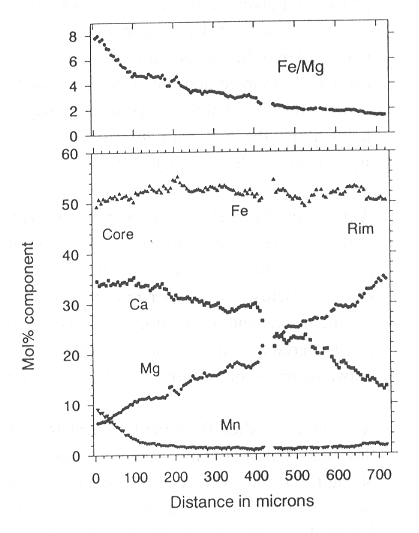 |
Fig. 3.4-2: Rim-rim zoning profile across an atoll garnet (left and right) as well as across a newly-formed garnet in the interior of a large phengite enclosed by the atoll. |
Fig. 3.4-3: Core-rim profile across a non-atoll grain of the same sample as for Fig. 3.4-2. |
A persistent problem in geothermobarometry remains the estimation of
the ratio of ferrous to ferric iron as the common tool used for mineral
analysis, the electron microprobe, cannot distinguish between these two.
For equilibria based on Fe2+-bearing phases, for example the
garnet-clinopyroxene Fe2+-Mg exchange geothermometer, failure
to subtract the ferric iron from total Fe can lead to a significant displacement
from the true equilibrium temperature. This problem is illustrated by another
Chinese eclogite sample where the main constituent phases, garnet and clinopyroxene,
have been analysed both by electron microprobe and by 57Fe Mössbauer
spectroscopy (in collaboration with D.A. Carswell, Sheffield and C. McCammon).
The Mössbauer spectrum for the clinopyroxene of this sample (Fig.
3.4-4) reveals that a considerable proportion of the iron, over 50%, occurs
as Fe3+. In contrast, only 6% of the iron in garnet of the same
sample is ferric. Ignoring the ferric iron in this sample results in a
distribution coefficient (KD) for Fe-Mg between garnet and clinopyroxene
of 6.13, yielding a temperature, at 2.5 GPa, of 840°C. Correcting for
the ferric iron in each phase increases KD to 12.5 and reduces
the calculated temperature to 650°C. Failure to recognise or to correct
for the effect of ferric iron clearly has a profound effect on geothermobarometric
results!
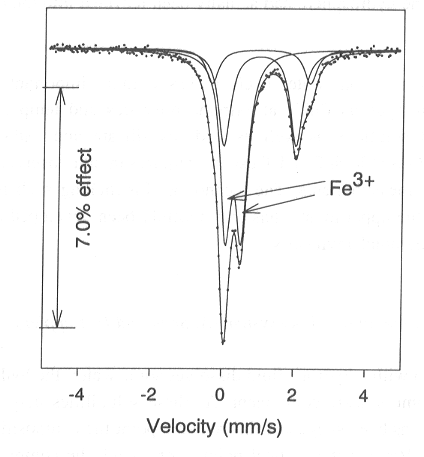 |
Fig. 3.4-4: Mössbauer spectrum for clinopyroxene from an eclogite. |

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page