

TiO2 is a minor component in the chemistry of the vast majority of Earth´s lavas. Still, the presence of TiO2 up to 11 wt% in natural liquids has been reported, and concentrations up to 16 wt% have been reported in lunar glasses. Furthermore, titanium has been demonstrated to have a strong effect on silicate melt properties, especially the molar volume, in contrast to the major elements occurring in ultramafic melts. We have therefore initiated experiments to measure one-atmosphere densities of a number of ultramafic Mare Ti-bearing melts at very high temperatures using the precise Ir-based double-bob Archimedean method. Samples were synthetic analogues of the Apollo 14 black glass, Apollo 15 green glass, Apollo 17 orange glass and a hypothetical lunar basalt, the differences between which are mainly the TiO2 and SiO2 contents. Densities, which are plotted in Fig. 3.6-10, were measured in the temperature range from 1773 K up to 2073 K and range from 3.01 to 2.73 g/cm3, the highest density corresponding to Ti-rich melt and the lowest to Ti-poor melt.
The present one-atmosphere measurements of these ultramafic Ti-bearing
melts exhibit some discrepancies with calculated values using existing
schemes. The difficulty in reproducing their one-atmosphere density likely
stems, at least in part, from the difficulties in constraining the partial
molar volume of the Ti component. Since titanium exhibits a dramatic effect
on the molar volume due to its structural specificity, investigations of
alkaline and earth-alkaline titanates are required in future studies for
a better determination of the partial molar volume of TiO2.
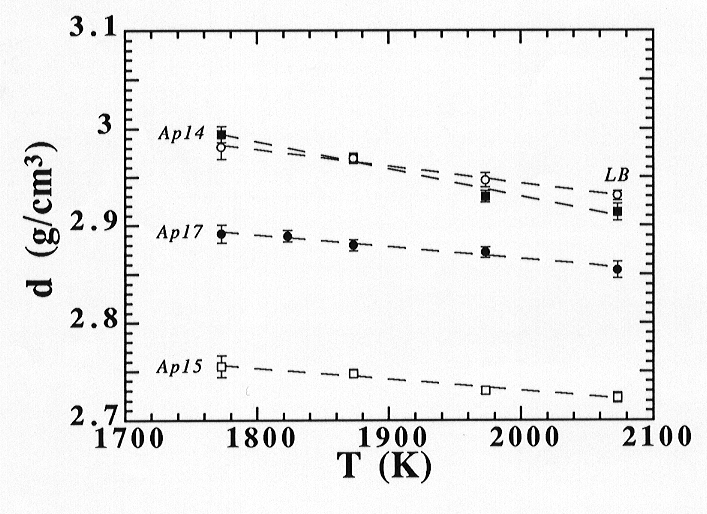 |
Fig. 3.6-10: One-atmosphere experimental densities of synthetic analogues of the Apollo 14 black glass (solid squares), Apollo 15 green glass (open squares), Apollo 17 orange glass (solid circles) and a hypothetical lunar basalt (open circles). |
Of more geologic significance, olivine crystal/ultrabasic liquid density inversions may have played an important role in the petrogenesis of ultramafic lunar magmas. In the lunar system, these crossovers may occur at relatively modest pressures because lunar magmas are significantly denser than their past and present terrestrial analogues, due to high concentrations of FeO and in some cases TiO2. Combined with high-pressure static experiments in progress at Harvard University on these lunar melts, the present one-atmosphere densities will yield their isothermal compression curves (i.e., new values of the isothermal bulk modulus and its pressure derivative through application of the third order Birch-Murnaghan equation of state). Thus, the density of ultramafic lunar magmas will likely place important constraints on their petrogenesis.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page