
Sergey V Ovsyannikov, Artem M Abakumov, Alexander A Tsirlin, Walter Schnelle, Ricardo Egoavil, Jo Verbeeck, Gustaaf Van Tendeloo, Konstantin V Glazyrin, Michael Hanfland & Leonid Dubrovinsky
Angewandte Chemie International Edition: DOI: 10.1002/anie.201208553, 2012.
http://onlinelibrary.wiley.com/doi/10.1002/anie.201208553/abstract
Manganites, MMnO3 (where M is a metal or combination of metals in dodecahedrally-coordinated by oxygen A-position) is one of the most important and industry-relevant sub-classes of perovskites revealed numerous exciting phenomena, including colossal magnetoresistance near the Curie temperature, dense granular magnetoresistance, optically-induced magnetic phase transitions, and many others. In manganites the Mn ions exclusively occupy the octahedrally-coordinated B-sites in the crystal structure. Moreover, so far there were no known M2O3 oxides adopting a perovskite structure. A new work performed at BGI (selected as a VIP – Very Important Paper in Angewandte Chemie and highlighted at the cover page of one of the issues) reports on a high-pressure and high-temperature (HP-HT) synthesis of two new polymorphs of manganese sesquioxide, Mn2O3 recoverable at ambient conditions, namely, corundum-type ε-Mn2O3 and perovskite-type ζ-Mn2O3. ζ-Mn2O3 is the first binary oxide adopting perovskite-type AA'3B4O12 crystal structure. It shows an unprecedented example of charge disproportionation that can be expressed as Mn2+(Mn3+)3(Mn3.25+)4O12 according to several distinct crystallographic positions of Mn. The proven existence of the ‘binary’ perovskite structure in a simple oxide advances our understanding of crystal chemistry of geo-materials at conditions relevant to the Earth interiors. Furthermore, this case shows that HP-HT treatment could masterly tune the oxidation states of the Mn ions to form new exotic structures, and hence suggests that new classes of perovskite manganites may be synthesized.
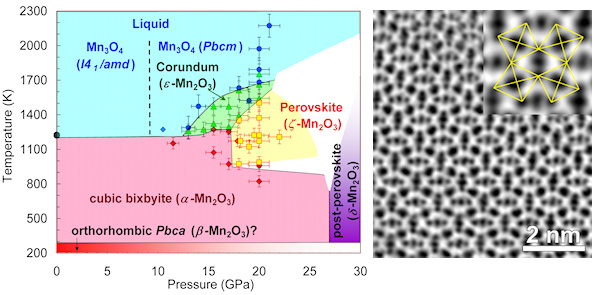
Pressure-temperature apparent phase diagram of Mn2O3 and ABF-STEM image of the perovskite-type ζ-Mn2O3 taken along the <100>p perovskite direction (the yellow lines trace tilted MnO6 octahedra)
Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de